![[Laboratory VII - The Origin of Seed Plants]](OspD/OspfontN.jpeg)
![[Laboratory VII - The Origin of Seed Plants]](OspD/OspfontN.jpeg)
Progymnosperms, as the name implies, are plants that share important derived features with seed plants but are still free-sporing themselves. They were important components of the flora from the Middle Devonian through the Mississippian (Lower Carboniferous in Europe). These plants commonly have secondary vascular tissues (a.k.a., wood) and some grew to be tall trees. However, they were spore-bearing. Some were homosporous like other Devonian euphyllophytes (e.g., Psilophyton and Pertica), while others were heterosporous (produced two types of spores).
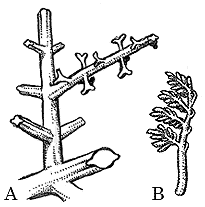
Tetraxylopteris. Tetraxylopteris from the Middle and Late Devonian resembles Pertica in some important ways, except branching pattern has become even more organized (Figure 7.4) (VG 1:3). Branching in Tetraxylopteris is decussate, meaning that pairs of branches emerge together on opposite sides of the main axis, with pairs above and below arranged at right angles to the pair between. In sterile axes, terminal branches have been interpreted as leaves. However, branching in these "leaves" is still very irregular and they are not webbed in with tissue as is the case with later forms like Archaeopteris.
| Figure 7.4: Tetraxylopteris. (A) Sterile branch -- note opposite decussate arrangement of appendages. (B) Fertile branch with sporangia on pinnate ultimate branches. |
|
Fertile axes of Tetraxylopteris (Figure 7.4) show complex branching like fertile stems, but ultimate portions are much more regular in branching pattern. Fertile branches arise in place of the leaf-like branch systems. Each sporangial complex is born on a branch system that dichotomizes twice, with each resulting branch tip branching pinnately and ending in a sporangium (Figure 7.4). Sporangia open laterally, a primitive feature retained from Pertica and Psilophyton-grade ancestors. However, Tetraxylopteris appears to have a zone of specialized cells to facilitate sporangial dehiscence. Tetraxylopteris was homosporous also like earlier euphyllophytes.
Aneurophyton. Aneurophyton is also known from the Middle and Late Devonian. Aneurophyton is similar to Tetraxylopteris in overall form, but its branches are arranged helically rather than decussately. As with Tetraxylopteris, the dichotomous ultimate branches in Aneurophyton are interpreted as "leaves" but they are similarly un-webbed and so this interpretation is generous. However, small terminal branches undoubtedly increased photosynthetic surface area (Figure 7.5a).

|
| Figure 7.5: Aneurophyton. (A) Portion of sterile shoot showing ultimate branches that have been interpreted as "leaves". (B) Fertile ultimate appendage showing recurved sporangia.
|
In fertile axes, ultimate branches are replaced by dichotomies with multiple homosporous sporangia arising along the length of each fork of the branch. Sporangia appear to recurve toward the center of the dichotomy (Figure 7.5b), but this may be an artifact of preservation. The main axis of Aneurophyton has a lobed protostele and produced a small amount of secondary xylem from meristematic regions between the lobes. Known axes are relatively slender suggesting a bush or vine growth form rather than a large tree.
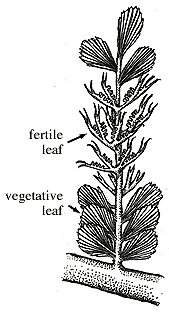
Archaeopteris. Archaeopteris (not Archaeopteryx!) is abundant from the Late Devonian through the Mississippian. The name Archaeopteris ("ancient fern") was applied to frond-like two-dimensional branch systems with terminal branches webbed in with photosynthetic tissue (Figure 7.6) (VG 1:4). In fertile examples, a branch bearing stalked pairs of elliptical sporangia replace webbed leaves. In some species, fertile branches occur on the same axis with photosynthetic leaflets; in others, whole branch systems are dedicated to reproduction.
|
Figure 7.6: Archaeopteris foliage showing webbed leaflets and fertile branches intermixed on the same axis. Woody stem is Callixylon. |
Webbed leaves and two-dimensional branching differentiate Archaeopteris and its relatives from related plants such as Aneurophyton and Tetraxylopteris. Heterospory is also derived in Archaeopteris. Trilete microspores are 33-70 mm in diameter; megaspores range from 110-500 mm were dispersed into the environment, suggesting a free-sporing life history.
The wood or Archaeopteris -- Callixylon(VG 1:5)(VG 1:6) -- was described in 1911 as Late Devonian conifer from Russia. Logs well over a meter in diameter and 10 m long are not uncommon, leading to the interpretation that they represented some of the first true trees (Figure 7.7) (VG 1:7). The interpretive error is understandable because Callixylon wood differs from that conifers by only a few details of the anatomy of its pits. However, it remains controversial whether wood is independently derived in progymnosperm and seed plant lineages.
The relationship between Callixylon wood and Archaeopteris foliage was not recognized until C.B. Beck happened upon a museum specimen of Archaeopteris that was attached to a small portion of the woody stem. By luck, the wood was preserved anatomically (permineralized). Upon sectioning the specimen, Beck recognized the wood as Callixylon. In 1960, Beck reported the organic connection between Archaeopteris and Callixylon and established a new and extinct group of plants -- the progymnosperms -- to contain woody plants with fern-like foliage and
reproduction.

|
| Figure 7.7: Reconstruction of tree-sized Archaeopteris. Note that the foliage name, Archaeopteris, applies to the whole plant because this name was given first.
|
The evolution of heterospory among the progymnosperms placed them at center stage in the debate over the origin of the seed habit. Many early workers argued that a heterosporous plant like Archaeopteris was the most likely ancestor to the seed plants because the gradualist paradigm of evolution required a series of morphological intermediates. According to this argument, heterospory was the intermediate between homosporous free-sporing reproduction and the retained endosporic gametophyte of the seed habit.
W.A. DiMichele and colleagues (1989) proposed that the seed habit could evolve directly from a homosporous ancestor like Aneurophyton by speeding up sexual maturation of the megagametophyte such that archegonia and eggs were produced before spores were dispersed. This hypothesis bypassed the free-sporing heterosporous ancestor. They also argued that the ecology of free-sporing heterospory -- typically plants growing in standing-water swamps -- was unlikely to have given rise to early seed plants, which were thought to have pioneered in well-drained upland habitats.
At the moment, both hypotheses remain widely discussed. Future fossil discoveries and more detailed phylogenetic analyses of the plants critical to seed plant evolution may help clarify the picture. However, recent discovered in plant developmental genetics have shown that dramatic changes in morphology, like those needed to evolve seeds from a homosporous aneurophyte, are possible. The linking of the apparently unrelated fields of paleobotany and developmental genetics may hold many important clues to this major evolutionary event -- the origin of the seed habit.
![[Previous Page]](../VPLimg/Back.jpeg) |
![[Title Page]](OspD/Ospbutt.jpeg) |
![[Glossary]](../VPLimg/Glossbutt.jpeg) |
![[Range Chart]](../VPLimg/Rangebutt.jpeg) |
![[Geologic Time Scale]](../VPLimg/timesbutt.jpeg) |
![[Next Page]](../VPLimg/Forward.jpeg) |

